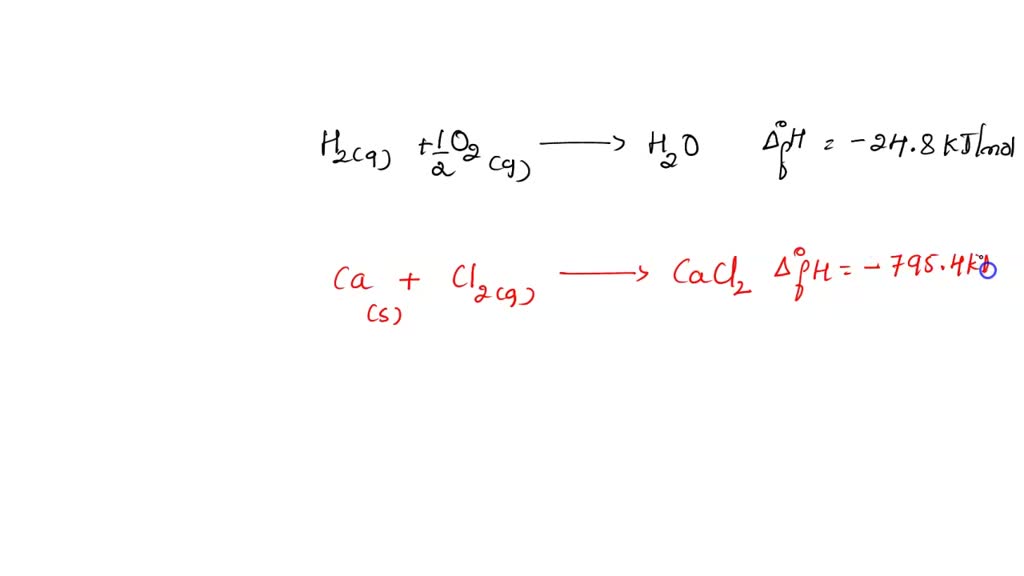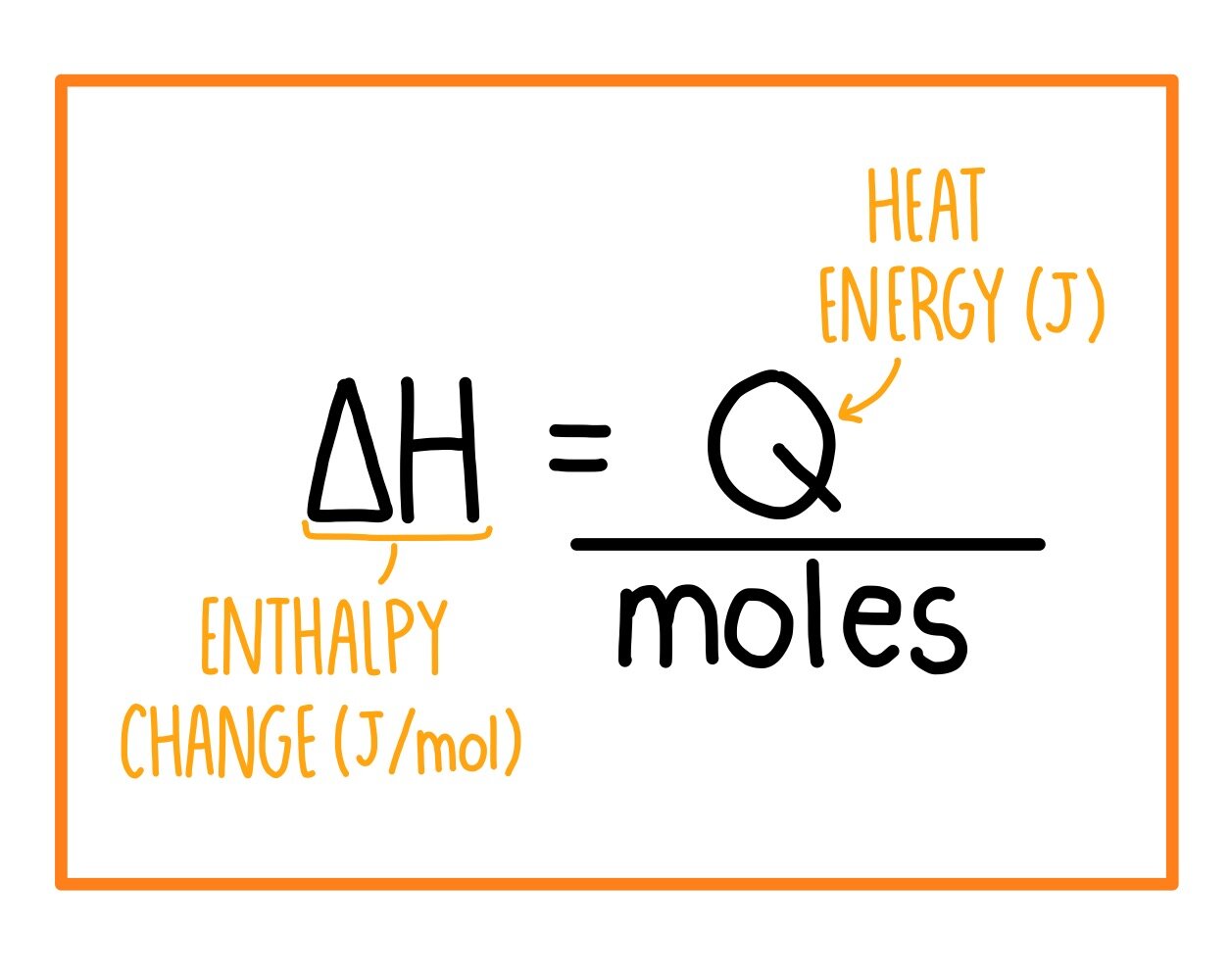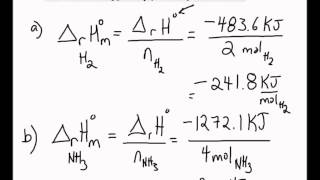Calculations in Chapter 10. Molar Enthalpy of Fusion Used when melting or freezing = ___energy ____ mol of substance Can be arranged to find any of the. - ppt download

SOLVED: I1. The standard molar enthalpy of formation of COz (g) at 298 K is -393.509 klhmol: Use data to calculate the value of AH? at 1000 K Assume the gases behave

3:04 calculate the molar enthalpy change (ΔH) from the heat energy change, Q - TutorMyself Chemistry